Diazinon is a nonsystemic organophosphate insecticide developed in 1952, used to control cockroaches, silverfish, ants, and fleas in residential, non-food buildings. It is used on home gardens and farms to control a wide variety of sucking and leaf eating insects. It is used on rice, fruit trees, sugarcane, corn, tobacco, potatoes and on horticultural plants. It is also an ingredient in pest strips. Diazinon has veterinary uses against fleas and ticks. It is available in dust, granules, seed dressings, wettable powder, and emulsifiable solution formulations.Toxic effects of diazinon are due to the inhibition of acetylcholinesterase, an enzyme needed for proper nervous system function. The range of doses that results in toxic effects varies widely with formulation and with the individual species being exposed. The toxicity of encapsulated formulations is relatively low because diazinon is not released readily while in the digestive tract. Some formulations of the compound can be degraded to more toxic forms. This transformation may occur in air, particularly in the presence of moisture, and by ultraviolet radiation. Most modern diazinon formulations in the U.K. are stable and do not degrade easily. The symptoms associated with diazinon poisoning in humans include weakness, headaches, tightness in the chest, blurred vision, nonreactive pinpoint pupils, salivation, sweating, nausea, vomiting, diarrhea, abdominal cramps, and slurred speech. Death has occurred in some instances from both dermal and oral exposures at very high levels.
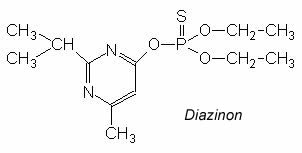
IUPAC Formula: O,O-diethyl O-2-isopropyl-6-methylpyrimidin-4-yl phosphorothioate
Chemical Formula: C12H21N2O3PS
LD50/LC50: The LD50 is 300 to 400mg/kg for technical grade diazinon in rats. The inhalation LC50 (4-hour) in rats is 3.5 mg/L In rabbits, the dermal LD50 is 3600 mg/kg.
Effects on Wildlife: Birds are quite susceptible to diazinon poisoning. The use of diazinon in open areas poses a "widespread and continuous hazard" to birds. Birds are significantly more susceptible to diazinon than other wildlife. LD50 values for birds range from 2.75 mg/kg to 40.8 mg/kg. Diazinon is highly toxic to fish. In rainbow trout, the diazinon LC50 is 2.6 to 3.2 mg/L. There is some evidence that saltwater fish are more susceptible than freshwater fish. Studies show that diazinon does not bioconcentrate significantly in fish.
***Diazinon is highly toxic to bees***.
Breakdown in soil and groundwater: Diazinon has a low persistence in soil. The half-life is 2 to 4 weeks. Bacterial enzymes can speed the breakdown of diazinon and have been used in treating emergency situations such as spills. Diazinon seldom migrates below the top half inch in soil, but in some instances it may contaminate groundwater. The pesticide was detected in 54 wells in California and in tap water in Ottawa, Canada, and in Japan.
Breakdown in water: The breakdown rate is dependent on the acidity of water. At highly acidic levels, one half of the compound disappeared within 12 hours while in a neutral solution, the pesticide took 6 months to degrade to one half of the original concentration.
Breakdown in vegetation: In plants, a low temperature and a high oil content tend to increase the persistence of diazinon. Generally the half-life is rapid in leafy vegetables, forage crops and grass. The range is from 2 to 14 days. In treated rice plants only 10% of the residue was present after 9 days. Diazinon is absorbed by plant roots when applied to the soil and translocated to other parts of the plant.
Physical Properties:
- Appearance: Diazinon is a colorless to dark brown liquid. It has a flashpoint of 180 F.
- Chemical Name: O,O-diethyl 0-2-isopropyl-6-methyl(pyrimidine-4-yl) phosphorothioate.
- CAS Number: 333-41-5
- Molecular Weight: 304.35
- Water Solubility: 40 mg/L @ 20 C
- Solubility in Other Solvents: petroleum ether v.s.; alcohol v.s.; benzene v.s.
- Melting Point: Decomposes @ >120 C
- Vapor Pressure: 0.097 mPa @ 20 C
- Partition Coefficient: Not Available
- Adsorption Coefficient: 1000 (estimated)
Sources: World Health Organisation and Oregon State University.