Bromadiolone: does not occur naturally. It is used as a rodenticide in urban and farm rodent control and acts by disrupting the normal blood clotting mechanisms causing an increased tendency to bleed. The rodenticidal properties of bromadiolone were reported in 1976. It is a second generation anticoagulant that is effective against rats and mice, including those resistant to first generation anticoagulants. It is used in the form of ready-to-use baits of low concentration containing 0.005% bromadiolone. Bromadiolone is a white to off-white powder. Its solubility in water is very low (less than 20 mg/litre at 20°C). It is slightly soluble in ethanol and ethyl acetate, and soluble in dimethylformamide. The flash-point temperature is 218°C.Bromadiolone is absorbed through the gastrointestinal tract, skin, and respiratory system. The major route of elimination in different species after oral administration is via the faeces. The liver is the main organ of accumulation and storage. Bromadiolone has been found in the liver as the unchanged parent compound. Elimination from the liver is biphasic with an initial rapid phase of 2-8 days and a slower phase with a half-life of 170 days. No data are available on the kinetics and metabolism of bromadiolone in humans.
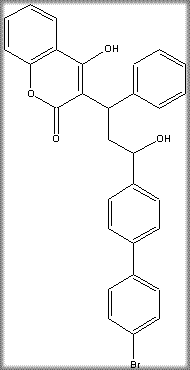
Chemical Name: 3-[3-(4′-bromobiphenyl-4-yl)-3-hydroxy-1-phenylpropyl]-4-hydroxycoumarin
Chemical Formula: C30H23BrO4
Chemical Structure:
LD50/LC50: Bromadiolone has a high, acute oral toxicity (LD50 of 1-3 mg/kg) for various species including rodents and non-rodents. The dermal toxicity is also high (LD50 of 9.4 mg/kg in rabbits). Signs of poisoning are those associated with an increased tendency to bleed. Bromadiolone is non-irritant to the skin. It is a slight irritant for the eye. In feeding studies on rats, the only effect found has been that associated with anticoagulant action. In a 12-week feeding study on rats, the maximum tolerated dose was 10 µg/kg body weight per day. Mutagenicity and teratogenicity studies have not shown any mutagenic, embryotoxic, or teratogenic effects.
Effects on Other Organisms in the Laboratory and Field: Bromadiolone has shown toxicity for aquatic organisms. The LC50 (96-h) for various fish species ranged from 1.4 to more than 3 mg/litre. Bird species appear to be less susceptible to bromadiolone than mammals with a reported acute, oral LD50 of at 138 mg/kg. Secondary poisoning through the consumption of rats and mice killed with bromadiolone may occur in dogs and cats in urban situations, but more likely in farm situations. As a technical material, bromadiolone is extremely toxic for many mammalian species. Signs of poisoning in all species, including humans, are associated with an increased tendency to bleed.
Evaluation of effects on the environment: Bromadiolone is applied to discrete sites in the form of low-concentration baits and is stable under normal conditions. Bromadiolone is poorly soluble in water and, in a bait formulation, it is is unlikely to be a source of water pollution. As a technical material, it is toxic for aquatic organisms.
Bromadiolone is readily adsorbed on soil, rich in clay and organic compounds, with no leaching; degradation in soil is significant. Non-target organisms are potentially at risk from direct consumption of baits (primary hazard) and through eating poisoned rodents (secondary hazard). Whole-grain baits are highly attractive to birds. Bird species appear to be less susceptible to bromadiolone than rodents.
The primary hazard is usually expressed by the amount of finished bait that must be consumed to approach the lethal dose. To reach the toxic or lethal dose, the non-target species must consume comparatively large amounts of bait with a concentration of 0.005% active ingredient. Some secondary toxicity laboratory studies on wildlife have shown that captive predators could be intoxicated by no-choice feeding of bromadiolone-poisoned or dosed prey. The significance of these results in terms of hazards under field conditions is difficult to assess, because the predators would not be expected to eat only poisoned animals. However, predators may take poisoned small mammals that are still alive, preferentially. In areas close to baiting, poisoned rodents may represent a high proportion of the diet for individual birds. However, only few individuals will be affected, unless there is very widespread and constant use of the baits. Therefore, some kills of owls can be expected, but there will be no severe population effects. This ties in with small numbers of poisoned owls observed in the field.